


We are (S)-Methyl 2-(3-(3-(2-(7-chloroquinolin-2-yl)vinyl)phenyl)-3-hydroxypropyl)benzoate CAS:142569-69-5 manufacturer and supplier in China, Pls send inquiry of to info@nbinno.com of visit www.nbinno.com our official website should you have any interests
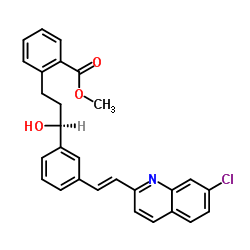
Chemical Name: (S)-Methyl 2-(3-(3-(2-(7-chloroquinolin-2-yl)vinyl)phenyl)-3-hydroxypropyl)benzoate
CAS.NO:142569-69-5
Synonyms:
Acid Methyl Ester [R-(E)-2-[3-[3-[2-(7-Chloro-2)
Benzoic acid, 2-[(3S)-3-[3-[(E)-2-(7-chloro-2-quinolinyl)ethenyl]phenyl]-3-hydroxypropyl]-, methyl ester
BENZOIC ACID,2-[3-(S)-[3-(E)-[2-(7-CHLORO-2-QUINOLINYL)ETHENYL]PHENYL]-3-HYDROXY
methyl 2-[3-(S)-[3-[2-(7-chloro-2-quiolinyl)ethenyl]-phenyl]-3-hydroxypropyl]benzoate
Molecular Formula:C28H24ClNO3
Molecular Weight:457.94800
Physical and Chemical Properties:
Density: 1.279 g / cm3
Boiling point: 643.972ºC at 760 mmHg
Melting point: 88-90ºC
Flash point: 343.262ºC
Refractive index: 1.668
Specification:
Appearance: Light yellow to yellow lumpy powder
Purity:≥98%
Heavy metals:≤20ppm
Water(KF):≤5%
Packing:
25kg cardboard drum or according to customer specified requirements
Storage:Stored in a cool and dry well-closed container. Keep away from moisture and strong light/heat.
Application:Intermediates of Montelukast Sodium CAS:151767-02-1
Intermediates of Montelukast CAS:158966-92-8
Related News: As a result, the Company’s platform is uniquely capable of overcoming numerous limitations associated with the production of cell therapies using patient- or donor-sourced cells, which is logistically complex and expensive and is subject to batch-to-batch and cell-to-cell variability that can affect clinical safety and efficacy.2235-46-3 Commonly used generic drugs: the products whose patents have expired and the generic drugs of many companies have participated in the competition, and the market demand is large.1-naphthol Commonly used generic drugs: the products whose patents have expired and the generic drugs of many companies have participated in the competition, and the market demand is large.6940-49-4 The intravenous form of rigosertib has been studied in Phase 1, 2, and 3 clinical trials involving more than 1000 patients, and is currently being evaluated in a randomized Phase 3 international INSPIRE trial for patients with HR-MDS after failure of HMA therapy.The intravenous form of rigosertib has been studied in Phase 1, 2, and 3 clinical trials involving more than 1000 patients, and is currently being evaluated in a randomized Phase 3 international INSPIRE trial for patients with HR-MDS after failure of HMA therapy.
| Product Name | |
|---|---|
| 2,3,5,6-Tetrafluorobenzyl alcohol | View Details |
| Palmatine Cas:3486-67-7 | View Details |
| Monoglyceride Cas:123-94-4 | View Details |