


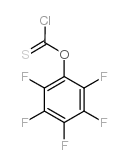
We are O-(2,3,4,5,6-pentafluorophenyl) chloromethanethioate CAS:135192-53-9 manufacturer and supplier in China, Pls send inquiry of to info@nbinno.com of visit www.nbinno.com our official website should you have any interests
Related News: Profitability continued to improve. The company’s comprehensive gross profit margin for the first three quarters was 36.06%, a year-on-year increase of 3.44 percentage points. Since this year, the company’s profitability has remained at a relatively high level. On the one hand, due to the decline in the trading business with lower profitability, the proportion has declined.Benzyl 3-oxoazetidine-1-carboxylate Profitability continued to improve. The company’s comprehensive gross profit margin for the first three quarters was 36.06%, a year-on-year increase of 3.44 percentage points. Since this year, the company’s profitability has remained at a relatively high level. On the one hand, due to the decline in the trading business with lower profitability, the proportion has declined.[1- (4-fenil-1H-imidazol-2-il) -etil] -carbámico éster bencílico del ácido CAS:864825-21-8 Profitability continued to improve. The company’s comprehensive gross profit margin for the first three quarters was 36.06%, a year-on-year increase of 3.44 percentage points. Since this year, the company’s profitability has remained at a relatively high level. On the one hand, due to the decline in the trading business with lower profitability, the proportion has declined.2-fluoro-4- (trifluorometil) benzaldehído CAS:89763-93-9 Pre-approval Access Programs (also known as expanded access, early access, compassionate use, named patient supply) are regulatory-compliant processes permitting experimental agents in development to be made available upon the request of a physician or a patient for appropriate patients for whom no alternative treatment option exists in their country.Pre-approval Access Programs (also known as expanded access, early access, compassionate use, named patient supply) are regulatory-compliant processes permitting experimental agents in development to be made available upon the request of a physician or a patient for appropriate patients for whom no alternative treatment option exists in their country.
| Product Name | |
|---|---|
| Rivaroxaban Cas:366789-02-8 | View Details |
| DL-LACTIC ACID Cas:598-82-3 | View Details |
| N-(4-Aminobenzoyl)Amino Ethanol Cas:54472-45-6 | View Details |