


We are L-Serine CAS:56-45-1 manufacturer and supplier in China, Pls send inquiry of to info@nbinno.com of visit www.nbinno.com our official website should you have any interests
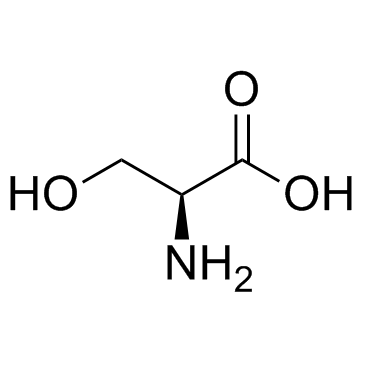
L-Serine
A nonessential amino acid in human. It is important in metabolism of purines and pyrimidines, and known as precursor of sphingolipids. It is widely used as an ingredient in infusion and infant formula.
| Cas.No | 56-45-1 |
| Assay | 99.0-101.0% |
| Specification | White crystals or crystalline powder, odorless, slightly sweet taste |
| Applications | Infusion |
| Packaging | 25kg&50kg |
| Pharmacopeia | JP,USP,EP,FCC |
| STORAGE | Controlled room temperature in tight container |
SPECIFICATION AND PROCEDURE
| State of solution (Transmittance) |
Not Less Than 98.0% |
| pH | 5.2~6.2 |
| Specific rotation[α]20D | +14.4~+15.5° |
| Specific rotation[α]25D | +14.0~+15.6° |
| Ammonium (NH4) | Not More Than 0.020% |
| Chloride (Cl) | Not More Than 0.020% |
| Sulfate (SO4) | Not More Than 0.020% |
| Iron (Fe) | Not More Than 10 ppm |
| Heavy metals (Pb)** | Not More Than 10 ppm |
| Arsenic (As2O3) | Not More Than 1 ppm |
| Loss on drying | Not More Than 0.20% |
| Residue on ignition | Not More Than 0.10% |
| Related substances | Not More Than 0.2% |
| Endotoxin* | Less Than 6.0 EU/g |
| Assay (dry basis) | 99.0~101.0% |
· * The endotoxin-certified grade will be supplied on request.
· ** FCC grade (Lead : Not More Than 5 mg/kg) will be supplied on request.
· This product meets requirements of residual solvents listed in the current JP, USP and EP.
Related News: Due to the cyclic nature of the upstream chemical products of the API, it is expected that the impact of price fluctuations of raw materials on the performance of API companies is unavoidable, but companies with thick product lines and the ability to produce intermediates are expected to minimize the upstream price impact.5-hydroxymethyl Tolterodine CAS:207679-81-0 Oligomannate, which uses extract from marine brown algae as raw material, received a conditional green light to treat mild-to-moderate level AD, the National Medical Products Administration (NMPA) said in a statement on its website late on Saturday.2-甲基-3-甲硫基呋喃 Onconova is currently in the clinical development stage with oral and IV rigosertib, including clinical trials studying single agent IV rigosertib in second-line higher-risk MDS patients (pivotal Phase 3 INSPIRE trial) and oral rigosertib plus azacitidine in first-line and refractory higher-risk MDS patients (Phase 2).Methyltriacetoxysilane “Administrative Measures for the Joint Review, Review and Approval of Raw Materials, Medicinal Auxiliaries and Pharmaceutical Packaging Materials and Pharmaceutical Preparations” (Consultation Draft) issued by the State Food and Drug Administration in December 2017. Supervision departments no longer accept applications for registration of APIs, pharmaceutical excipients, and packaging materials.Active Pharmaceutical Ingredients (APIs): Pharmaceutical active ingredients, which are the basic substances that constitute the pharmacological effects of pharmaceuticals, and are prepared by chemical synthesis, plant extraction, or biotechnology.
| Product Name | |
|---|---|
| 3-Amino-5-Chlorobenzotrifluoride | View Details |
| 8-fluoro-1,3,4,5-tetrahydro-azepino[5,4,3-cd]indol-6-one | View Details |
| cyclopentyl methyl ether Cas:5614-37-9 | View Details |