


We are Anilino-methyl-triethoxysilane CAS:3473-76-5 manufacturer and supplier in China, Pls send inquiry of to info@nbinno.com of visit www.nbinno.com our official website should you have any interests
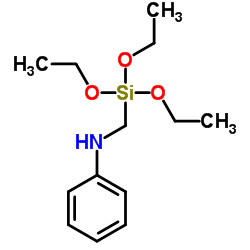
Chemical Name:Anilino-methyl-triethoxysilane
CAS.NO:3473-76-5
Synonyms:SILANE COUPLING AGENT ND-42;
N-PHENYLAMINOMETHYLTRIETHOXYSILANE; Anilinomethyl-triethoxy-silan;
AnilinoMethyltriethoxysliane; Phenylaminomethyltriethoxysilane;
Molecular Formula:C13H23NO3Si
Molecular Weight:269.41200
Physical and Chemical Properties:
Density:1.004;
Boiling point:132-142oC;
Flash point:100oC;
Index of Refraction:1.4857;
Specification:
Appearance:Colorless to yellowish clear liquid
Purity(GC):≥97.0%
Packing:200 kg/drum, can also be packaged according to customer requirements
Storage:Store in a cool, ventilated place
Application:It can be used in the production of silyl modified polymers which serve as binders in adhesives and sealants.
It also can be used as a crosslinker, water scavenger and adhesion promoter in silane-crosslinking formulations, such as adhesives, sealants and coatings.
It can be used as surface modifier for fillers (like glass, metal oxides, aluminum hydroxide, kaolin, wollastonite, mica) and pigments.
Related News: The outlook for a cure is clouded with theoretical uncertainties and high-profile failures. Pharmaceutical giants including Johnson & Johnson, Merck and Pfizer have ditched their projects on unsatisfactory data.Ácido trifluorometanosulfónico CAS:1493-13-6 Only when the drug substance is processed into a pharmaceutical preparation can it become a drug for clinical application.2-etil-5-metilpirazina CAS:13360-64-0 DMF is the main management method for APIs in developed countries in Europe and the United States. Under the DMF system, API companies can submit DMF filing documents to the regulatory authority at any time, but the regulatory authority will not conduct technical reviews on them. When the drug is administered, the regulatory authority will associate and review the drug substance and the preparation.N-(2-chloropyrimidin-4-yl)-2,3-dimethylindazol-6-amine At the same time, through years of imitation and advanced technology learning, more and more domestic companies have participated in the field of highly original and characteristic APIs.Many branded versions of drugs are currently more expensive in China than in other major markets. They could now be subjected to a centralized procurement program where manufacturers will have to go through a bidding process to get the right to supply drugs to public hospitals, the National Health Commission said in a document published on late Friday.
| Product Name | |
|---|---|
| 4-Amino-3-Bromo-5-Nitropyridine Cas:89284-05-9 | View Details |
| methyllithium Cas:917-54-4 | View Details |
| Diethyl ethoxymethylenemalonate | View Details |