


We are 4,4-Difluoro-N-((1S)-3-oxo-1-phenylpropyl)cyclohexanecarboxamide CAS:376348-78-6 manufacturer and supplier in China, Pls send inquiry of to info@nbinno.com of visit www.nbinno.com our official website should you have any interests
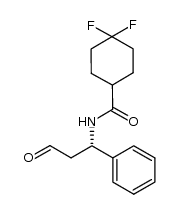
Product Name: 4,4-Difluoro-N-((1S)-3-oxo-1-phenylpropyl)cyclohexanecarboxamide
Synonym: 4,4-Difluorocyclohexanecarboxylic acid N-((1S)-3-oxo-1-phenylpropyl)amide; (S)-4,4-difluoro-N-(3-oxo-1-phenylpropyl)cyclohexanecarboxamide; Intermediate of Maraviroc
Cas No.: 376348-78-6
Formula: C16H19F2NO2
MW: 295.33
EINECS No.: N.A
HS Code: 2903890090
UN No.: N.A
Hazard class: Non-dangerous goods
Specification
| Items of Analysis | Standard of Analysis | Test Results |
| Appearance | White or similar white powder | Conforms |
| Purity (HPLC) | ≥99% | 99.75% |
| Single Impurity | ≤1.0% | 0.07% |
| Total impurities | ≤1.0% | 0.25% |
| Loss on drying | ≤0.5% | 0.12% |
| Residue on Ignition | ≤0.15% | 0.13% |
| Conclusion | Conforms to Factory Standard | |
Application
Intermediate of Maraviroc.
Packaging
25 kg/barrel, can also be packaged according to customer requirements.
Storage
Store in a cool, ventilated warehouse.
Keep away from fire and heat.
The temperature should not exceed 37 °C.
Related News: According to statistics, in 2017, China’s total production of chemical drugs reached 3.478 million tons, a year-on-year increase of 1.6%. The main business income showed a steady increase, from 328.972 billion yuan in 2012 to 573.475 billion yuan in 2017. The total profit was 48.644 billion yuan, but profit margins remain low (8.48% in 2017).1,8-Dibromooctane CAS:4549-32-0 At present, the additional 300 tons of sunniamine, 300 tons of fluoxamic acid, and 200 tons of cyprodinil have been successfully put into production, and the profitability is good.tenofovir hydrate At present, the additional 300 tons of sunniamine, 300 tons of fluoxamic acid, and 200 tons of cyprodinil have been successfully put into production, and the profitability is good.5-Bromoquinazolin-6-ylthiourea CAS:842138-74-3 It is understood that at present, the industry competition for global characteristic APIs has shown a development trend of vertical integration, and the number of mergers and acquisitions between pharmaceutical manufacturers and API manufacturers is increasing.The unit of the British drugmaker, which is challenging HIV drug market leader Gilead Sciences, said it will work with the FDA to determine the next steps for the new drug application.
| Product Name | |
|---|---|
| 2,4,5-Trifluorophenyl Acetic Acid Cas:209995-38-0 | View Details |
| 3-(1,1-diMethylethyl)dihydro-1,5-dioxo-(3R,7aR)-1H,3H-Pyrrolo[1,2-c]oxazole-7a(5H)-carboxaldehyde Cas:1214741-19-1 | View Details |
| di-tert-butyl peroxide Cas:110-05-4 | View Details |