


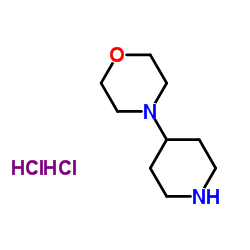
We are 4-Piperidin-4-ylmorpholine CAS:53617-35-9 manufacturer and supplier in China, Pls send inquiry of to info@nbinno.com of visit www.nbinno.com our official website should you have any interests
Related News: The oral form of rigosertib was developed to provide a potentially more convenient dosage form for use where the duration of treatment may extend to multiple years.3,5-Difluoro-2-methylbenzonitrile In recent years, China’s bulk drug companies have gradually completed the upgrade of the product structure of bulk raw materials to specialty raw materials and intermediates. The industry’s leading companies have further developed the research and development layout of high-barrier generic pharmaceutical raw materials with multiple patents that have not yet expired.4,4-Difluoropiperidine hydrochloride Beneficial drug raw materials refer to the active pharmaceutical ingredients used in the manufacture of original research drugs (patent drugs). They are mainly used to meet the needs of original multinational pharmaceutical companies and emerging biopharmaceutical companies for innovative drugs in clinical drug research, registration approval and commercialization sales , Which also contains advanced intermediates used in the manufacture of the drug substance that need to be regulated by regulatory authorities.(S)-tert-Butyl (4-chloro-3-oxo-1-phenylbutan-2-yl)carbamate CAS:102123-74-0 We are pleased that intravenous rigosertib will be made compliantly available to suitable patients with higher-risk MDS through their physicians in designated countries.Judging from the geographical distribution of enterprises, the largest distribution of API companies is Jiangsu and Zhejiang, with more than 300 enterprises; followed by Shandong, Sichuan and Hubei.
| Product Name | |
|---|---|
| Guanosine-9-17N, N-(2-methyl-1-oxopropyl)-, 2′,3′-diacetate Cas:163586-86-5 | View Details |
| 2-Trifluoromethylbenzonitrile Cas:447-60-9 | View Details |
| 1-(Tetrahydropyran-2-yl)-1H-pyrazole-5-boronic Acid Pinacol Ester Cas:903550-26-5 | View Details |