


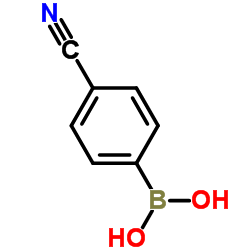
We are 4-Cyanophenylboronic acid CAS:126747-14-6 manufacturer and supplier in China, Pls send inquiry of to info@nbinno.com of visit www.nbinno.com our official website should you have any interests
Related News: We purchase the raw materials from raw material or chemical product manufacturers both in Japan and internationally. Clorhidrato de ácido (2-metilpiridin-4-il) borónico CAS:861905-97-7 This dosage form may also support combination therapy modalities.? To date, over 400 patients have been dosed with the oral formulation of rigosertib in clinical trials.? Combination therapy of oral rigosertib with azacitidine, the standard of care in HR-MDS, has also been studied. Currently, oral rigosertib is being developed as a combination therapy together with azacitidine for patients with higher-risk MDS who require HMA therapy.?1-(4-Hydrazinophenyl)methyl-1,2,4-triazole CAS:212248-62-9 This dosage form may also support combination therapy modalities.? To date, over 400 patients have been dosed with the oral formulation of rigosertib in clinical trials.? Combination therapy of oral rigosertib with azacitidine, the standard of care in HR-MDS, has also been studied. Currently, oral rigosertib is being developed as a combination therapy together with azacitidine for patients with higher-risk MDS who require HMA therapy.?HC VIOLET NO. 2 CAS:104226-19-9 This dosage form may also support combination therapy modalities.? To date, over 400 patients have been dosed with the oral formulation of rigosertib in clinical trials.? Combination therapy of oral rigosertib with azacitidine, the standard of care in HR-MDS, has also been studied. Currently, oral rigosertib is being developed as a combination therapy together with azacitidine for patients with higher-risk MDS who require HMA therapy.?In recent years, China’s bulk drug companies have gradually completed the upgrade of the product structure of bulk raw materials to specialty raw materials and intermediates. The industry’s leading companies have further developed the research and development layout of high-barrier generic pharmaceutical raw materials with multiple patents that have not yet expired.
| Product Name | |
|---|---|
| Triethoxysilane | View Details |
| 1-Bromononane | View Details |
| isovaleraldehyde Cas:590-86-3 | View Details |