


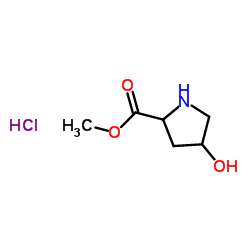
We are (2S,4R)-Methyl 4-hydroxypyrrolidine-2-carboxylate hydrochloride CAS:40216-83-9 manufacturer and supplier in China, Pls send inquiry of to info@nbinno.com of visit www.nbinno.com our official website should you have any interests
Chemical Name: (2S,4R)-Methyl 4-hydroxypyrrolidine-2-carboxylate hydrochloride
CAS.NO:40216-83-9
Synonyms:
trans-4-Hydroxy-L-proline Methyl Ester Hydrochloride
L-4-Hydroxyproline methyl ester hydrochloride
Methyl (2S,4R)-4-hydroxypyrrolidine-2-carboxylate
Methyl (2S,4R)-4-Hydroxypyrrolidine-2-carboxylate Hydrochloride
Molecular Formula: C6H12ClNO3
Molecular Weight:181.61700
Physical and Chemical Properties:
Density: /
Boiling point: 247.2ºC at 760 mmHg
Melting point: /
Flash point:103.3ºC
Refractive index: /
Specification:
Appearance: White powder
Purity:≥98%
Residue on Ignition: 0.10% max
Packing:
25kg cardboard drum or according to customer specified requirements
Storage:2-8ºC.Stored in a cool and dry well-closed container. Keep away from moisture and strong light/heat.
Application:Intermediates of Teneligliptin CAS:760937-92-6
Related News: Fate Therapeutics is a clinical-stage biopharmaceutical company dedicated to the development of first-in-class cellular immunotherapies for cancer and immune disorders.52605-97-7 Green Valley said it would launch the drug “very soon” in China. The company also aims to roll out a phase-3 clinical trial with sites in the United States, Europe and Asia in early 2020 to facilitate global regulatory approval of the drug.99-57-0 Green Valley said it would launch the drug “very soon” in China. The company also aims to roll out a phase-3 clinical trial with sites in the United States, Europe and Asia in early 2020 to facilitate global regulatory approval of the drug.3,5-dicloropiridina-2-carbonitrilo CAS:85331-33-5 This dosage form may also support combination therapy modalities.? To date, over 400 patients have been dosed with the oral formulation of rigosertib in clinical trials.? Combination therapy of oral rigosertib with azacitidine, the standard of care in HR-MDS, has also been studied. Currently, oral rigosertib is being developed as a combination therapy together with azacitidine for patients with higher-risk MDS who require HMA therapy.?This dosage form may also support combination therapy modalities.? To date, over 400 patients have been dosed with the oral formulation of rigosertib in clinical trials.? Combination therapy of oral rigosertib with azacitidine, the standard of care in HR-MDS, has also been studied. Currently, oral rigosertib is being developed as a combination therapy together with azacitidine for patients with higher-risk MDS who require HMA therapy.?
| Product Name | |
|---|---|
| Diosgenin | View Details |
| 4-Methylenepiperidine HCl Cas:144230-50-2 | View Details |
| Pyrazino[1′,2′:1,6]pyrido[3,4-b]indole-1,4-dione,6-(1,3-benzodioxol-5-yl)-2,3,6,7,12,12a-hexahydro-2,12a-dimethyl-, (6R,12aR)- Cas:NO: 477978-84-0 | View Details |