


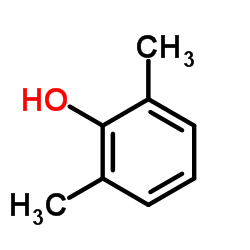
We are 2,6-Dimethylphenol CAS:576-26-1 manufacturer and supplier in China, Pls send inquiry of to info@nbinno.com of visit www.nbinno.com our official website should you have any interests
Related News: In 2016, the export volume of China’s specialty drug substances reached US $ 3.53 billion, accounting for 13.8% of the total drug substances.N4-Isobutylquinoline-3,4-diamine CAS:99010-09-0 In 2016, the export volume of China’s specialty drug substances reached US $ 3.53 billion, accounting for 13.8% of the total drug substances.γ-Butyrolactone CAS:96-48-0 Exceeding impurities in the drug substance may cause the product and its preparation to be recalled. The company receives an FDA warning letter or a CEP certificate suspension, which in turn will cause customer compensation, product recall costs, and asset impairment losses to affect the company’s performance.Piridoxal-5-fosfato CAS:54-47-7 The outlook for a cure is clouded with theoretical uncertainties and high-profile failures. Pharmaceutical giants including Johnson & Johnson, Merck and Pfizer have ditched their projects on unsatisfactory data.Only when the drug substance is processed into a pharmaceutical preparation can it become a drug for clinical application.
| Product Name | |
|---|---|
| hydrochlorothiazide | View Details |
| ALLYL CYCLOHEXYLPROPIONATE Cas:2705-87-5 | View Details |
| (1S,3S,5S)-tert-Butyl 3-carbamoyl-2-azabicyclo[3.1.0]hexane-2-carboxylate | View Details |