


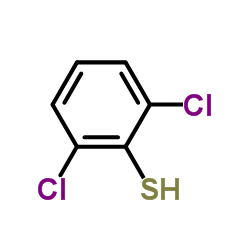
We are 2,6-dichlorobenzenethiol CAS:24966-39-0 manufacturer and supplier in China, Pls send inquiry of to info@nbinno.com of visit www.nbinno.com our official website should you have any interests
Related News: The purpose of APIs according to the FDA is to cause ‘pharmacological activity or other direct effects in the diagnosis, cure, mitigation, treatment or prevention of disease or to affect the structure and function of the human body.2,8-dibromodibenzofuran The purpose of APIs according to the FDA is to cause ‘pharmacological activity or other direct effects in the diagnosis, cure, mitigation, treatment or prevention of disease or to affect the structure and function of the human body.123-54-6 Green Valley said it would launch the drug “very soon” in China. The company also aims to roll out a phase-3 clinical trial with sites in the United States, Europe and Asia in early 2020 to facilitate global regulatory approval of the drug.2-Methyl-1-butanethiol CAS:1878-18-8 Green Valley said it would launch the drug “very soon” in China. The company also aims to roll out a phase-3 clinical trial with sites in the United States, Europe and Asia in early 2020 to facilitate global regulatory approval of the drug.This dosage form may also support combination therapy modalities.? To date, over 400 patients have been dosed with the oral formulation of rigosertib in clinical trials.? Combination therapy of oral rigosertib with azacitidine, the standard of care in HR-MDS, has also been studied. Currently, oral rigosertib is being developed as a combination therapy together with azacitidine for patients with higher-risk MDS who require HMA therapy.?
| Product Name | |
|---|---|
| Platinum powder Cas:7440-06-4 | View Details |
| Phenyltrimethoxysilane | View Details |
| Tert-butyl 4-(4-iodo-1H-pyrazol-1-yl)piperidine-1-carboxylate Cas:877399-73-0 | View Details |