


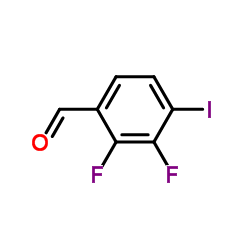
We are 2,3-Difluoro-4-iodobenzaldehyde CAS:885590-99-8 manufacturer and supplier in China, Pls send inquiry of to info@nbinno.com of visit www.nbinno.com our official website should you have any interests
Related News: The company’s short-term plan is to move from a purely chemical intermediate manufacturer to a formula active material supplier, and its long-term goal is to become a solution provider in the selected segment. In the future, this business is expected to become a new growth point for the company’s profit.Acetato de retinilo CAS:127-47-9 Oligomannate, which uses extract from marine brown algae as raw material, received a conditional green light to treat mild-to-moderate level AD, the National Medical Products Administration (NMPA) said in a statement on its website late on Saturday.2,3-Difluoro-6-nitroaniline Oligomannate, which uses extract from marine brown algae as raw material, received a conditional green light to treat mild-to-moderate level AD, the National Medical Products Administration (NMPA) said in a statement on its website late on Saturday.6-Bromo-2,3-Dihydro-4H-Chromen-4-One Pharmaceutical intermediates: chemical raw materials or chemical products used in the process of pharmaceutical synthesis, are intermediate products in the process of producing APIs, and can be further processed into APIs.“Administrative Measures for the Joint Review, Review and Approval of Raw Materials, Medicinal Auxiliaries and Pharmaceutical Packaging Materials and Pharmaceutical Preparations” (Consultation Draft) issued by the State Food and Drug Administration in December 2017. Supervision departments no longer accept applications for registration of APIs, pharmaceutical excipients, and packaging materials.
| Product Name | |
|---|---|
| N-methylmorpholine Cas:109-02-4 | View Details |
| 4-Chloro-6,7-bis(2-methoxyethoxy)quinazoline | View Details |
| Trans-Bis(Benzonitrile)Dichloropalladium(Ii) Cas:14220-64-5 | View Details |