


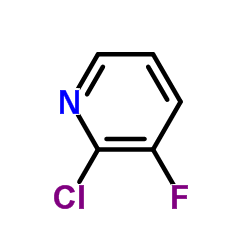
We are 2-Chloro-3-fluoropyridine CAS:17282-04-1 manufacturer and supplier in China, Pls send inquiry of to info@nbinno.com of visit www.nbinno.com our official website should you have any interests
Related News: In the context of the new medicine policy, with the development of China’s R & D strength, the number of innovative drugs approved has been blown out, which further strengthened the demand and speed of industrialization of raw materials in China.2-Bromophenylboronicacid CAS:244205-40-1 The drug substance is an active product that has completed the synthetic route, and the intermediate is a product in one place in the synthetic route.2,4,6-trihidroxibenzaldehído CAS:487-70-7 In 2004, the National Development and Reform Commission, the former Ministry of Health, and the Ministry of Finance jointly formulated and promulgated the Administrative Measures for the Configuration and Use of Large Medical Equipment. This method uses X-ray electronic computer tomography (CT) devices and medical magnetic resonance imaging equipment, digital subtraction angiography X-ray machines of 800 mA or more as the management products of large-scale medical equipment, Purchase and use need to be reported to the provincial health administrative department for approval before approval. However, in recent years, with the comprehensive coverage of medical insurance, such basic medical facilities are being widely popularized, and the relevant implementation rules have directly linked the number of equipment to the number of doctors. Therefore, as the demand for imaging equipment continues to increase, it will have a greater boosting effect on the upstream contrast agent and its raw material market.2-acetyl-5-methylfuran CAS:1193-79-9 The Breakthrough Designation for the iLet Bionic Pancreas System contemplates configurations with most insulin analogs approved for pumping as well as dasiglucagon, Zealand Pharma’s stable pumpable glucagon analog, which has a unique stability profile in a ready-to-use aqueous solution.The Company’s immuno-regulatory product candidates include ProTmune, a pharmacologically modulated, donor cell graft that is currently being evaluated in a Phase 2 clinical trial for the prevention of graft-versus-host disease, and a myeloid-derived suppressor cell immunotherapy for promoting immune tolerance in patients with immune disorders. Fate Therapeutics is headquartered in San Diego, CA.
| Product Name | |
|---|---|
| 2-(2H-Benzotriazol-2-yl)-p-cresol | View Details |
| O-(6-Chlorobenzotriazol-1-yl)-N,N,N’,N’-tetramethyluronium Tetrafluoroborate | View Details |
| 1,1-diphenyl-4-piperidin-1-ylbutan-1-ol,hydrochloride Cas:3254-89-5 | View Details |