


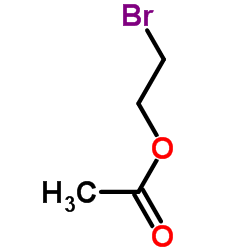
We are 2-Bromoethyl acetate CAS:927-68-4 manufacturer and supplier in China, Pls send inquiry of to info@nbinno.com of visit www.nbinno.com our official website should you have any interests
Related News: “Once these risks have been identified then a project team can be put together combining the necessary expertise to provide the integrated services and tailored support to ensure continuity of supply from beginning to end.”2-(tert-Butylamino)acetic acid hydrochloride Characteristic APIs represented by cardiovascular, antiviral, antitumor and other categories are targeted at generic APIs with patent expiring original research drugs, which have high scientific and technological content and rich profits. They have developed in China in the past decade. The more active API segment has contributed to the rise of Huahai Pharmaceutical, Chuangnuo Pharmaceutical, and Jiangbei Pharmaceutical.110-52-1 Because API companies usually adopt a different synthetic route from the original research company, compared with the drug itself, the impurities contained in the generic drug API have not been verified by the original drug for many years of use and require more careful testing and control.126325-50-6 This is of vital important and underlies one of the most solid principles and regulations of modern pharmacy.DMF is the main management method for APIs in developed countries in Europe and the United States. Under the DMF system, API companies can submit DMF filing documents to the regulatory authority at any time, but the regulatory authority will not conduct technical reviews on them. When the drug is administered, the regulatory authority will associate and review the drug substance and the preparation.
| Product Name | |
|---|---|
| 1,4-difluorobenzene | View Details |
| THIABENDAZOLE Cas:148-79-8 | View Details |
| 4-Amino-3-methoxybenzoic Acid Cas:2486-69-3 | View Details |