


We are 2-(7-Methoxy-3,4-dihydronaphthalen-1-yl)acetonitrile CAS:861960-34-1 manufacturer and supplier in China, Pls send inquiry of to info@nbinno.com of visit www.nbinno.com our official website should you have any interests
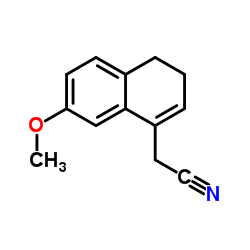
Chemical Name: 2-(7-Methoxy-3,4-dihydronaphthalen-1-yl)acetonitrile
CAS.NO: 861960-34-1
Synonyms:1-Naphthaleneacetonitrile, 3,4-dihydro-7-methoxy-
(7-Methoxy-3,4-dihydro-1-naphthalenyl)acetonitrile
Molecular Formula: C13H13NO
Molecular Weight: 199.24800
Physical and Chemical Properties:
Density: 1.092 g/cm3
Boiling point:349.603ºC at 760 mmHg
Melting point: /
Flash point:147.517ºC
Refractive index: 1.555
Specification:
Appearance: Pale yellow crystals
Purity:≥99.0%
Packing:
25kg 1 cardboard drum or according to customer specified requirements
Storage:Store in a cool, dry place. Store in a tightly closed container.
Application:Intermediates of Agomelatine CAS:138112-76-2.
Related News: The chemical compound which is in the process of becoming an API from a raw material is called an intermediate.5-Bromo-2-methoxy-3-nitropyridine With the improvement of domestic GMP management level, the increase of process development capabilities and international certification experience, China has already possessed the conditions for developing characteristic APIs.5-amino-1-(N-methylcarbamoyl)imidazole-4-carboxamide In recent years, with the increasing number of patent medicines whose patents have expired, the variety and quantity of generic drugs have also increased rapidly, which has brought huge market opportunities to the API market and the output of APIs has continued to increase.1-β-D-Arabinofuranosiluracilo CAS:3083-77-0 Rigosertib is a small molecule that inhibits cellular signaling in cancer cells by acting as a RAS mimetic. Current clinical development of rigosertib is centered upon the therapeutic management of MDS, a heterogeneous group of bone marrow disorders characterized by ineffective hematopoiesis that often develop into acute myeloid leukaemia (AML).ViiV, in which Pfizer and Shionogi have small stakes, said it received a so-called complete response letter (CRL) from the FDA in which the regulator questioned the treatment’s chemistry, manufacturing and controls process, but not its safety.
| Product Name | |
|---|---|
| 2,2-DIMORPHOLINODIETHYLETHER (DMDEE) Cas:6425-39-4 | View Details |
| 2,2′-DIHYDROXY-4-METHOXYBENZOPHENONE Cas:131-53-3 | View Details |
| 2-Chlorophenol Cas:95-57-8 | View Details |