


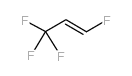
We are 1,3,3,3-TETRAFLUOROPROP-1-ENE CAS:1645-83-6 manufacturer and supplier in China, Pls send inquiry of to info@nbinno.com of visit www.nbinno.com our official website should you have any interests
Market News: UQUIFA is a manufacturer of APIs and intermediates for the pharmaceutical industry globally. The company has 3 manufacturing sites (2 in Spain and 1 in Mexico) with strict adherence to all regulatory norms. pyridine-4-sulfonic acid salicylidenehydrazide manufacturer.The company will see an immediate $6 billion bump to its topline thanks to Alexion’s C5 inhibitors, used to treat rare diseases like paroxysmal nocturnal hemoglobinuria. 1-(6-(4-ethoxyphenyl)-3-(methylthio)-5,6-dihydro-7H-benzo[b][1,2,4]triazino[6,5-d]azepin-7-yl)ethan-1-one supplier Events, Hatzalah of South Florida, and Ygrene and in partnership with the Shul of Bal Harbour we are able to continue this mission by providing first and last month rent as well as the security deposit for these families If this fund grows bigger, we will be able to continue to contribute funds directly for the rents.” Michael Capponi
“During this challenging time for the survivors, the uncertainty of not knowing where they will live exacerbates an already extremely painful and tragic situation. We are committed to the relocation efforts of all of the survivors through our partnership with Global Empowerment Mission.” Rabbi Zalman Lipskar, The Shul of Bal Harbour.
“We are devastated by the loss suffered by the victims, families of victims and the community at the hands of the Surfside tragedy. It is an honor to contribute to GEM’s Champlain Survivor Relocation Fund to provide safe and comfortable homes for survivors to help them get back on their feet, ” said Ygrene CEO, Jim Reinhart.
Baclofen is a muscle relaxant drug indicated for the treatment of muscle pain, spasms, and stiffness in people with multiple sclerosis, spinal cord injury, or disease.
This is Beximco Pharma’s ninth Abbreviated New Drug Application fully developed in-house and successfully approved for the US market since the Company’s oral solid dosage facility was approved by the US FDA in June 2015.
According to IQVIA audited market data, there are currently nine active players for Baclofen in the US market, which generated sales of more than $110 million in 2020.
Mr. Nazmul Hassan MP, Managing Director of Beximco Pharmaceuticals, said: We are pleased to have received FDA approval of Baclofen, a commonly prescribed medicine in the US, as we continue to leverage our core strengths in R&D and manufacturing to develop and deliver important generic products to patients. Phosphonic acid, [[(4-methylphenyl)amino](4-nitrophenyl)methyl]- producer
| Product Name | |
|---|---|
| 11-(1-Piperazinyl)-Dibenzo[b,f][1,4]Thiazepine Dihydrochloride | View Details |
| 2,4,6-Trifluorophenol | View Details |
| 2,8-dibromodibenzofuran | View Details |