


We are 1,2-Bis(triethoxysilyl)ethane CAS:16068-37-4 manufacturer and supplier in China, Pls send inquiry of to info@nbinno.com of visit www.nbinno.com our official website should you have any interests
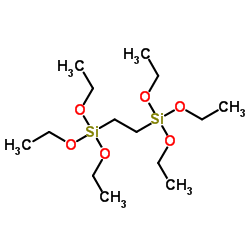
Chemical Name:1,2-Bis(triethoxysilyl)ethane
CAS.NO:16068-37-4
Synonyms:Ethylenebis(triethoxysilane); triethoxy(2-triethoxysilylethyl)silane; 4,4,7,7-Tetraethoxy-3,8-dioxa-4,7-disiladecane;
Molecular Formula:C14H34O6Si2
Molecular Weight:354.58700
Physical and Chemical Properties:
Density:0.958;
Melting point:-30oC;
Boiling point:119oC;
Flash point:113oC;
Index of Refraction:1.411;
Specification:
Appearance:Clear to straw liquid
Purity(GC):≥98.0%
Packing:190 kg/drum, can also be packaged according to customer requirements
Storage:Store in a cool, ventilated place
Application:It can be used to improve the adhesion of RTV silicone sealants to dry or wet surface, and improve its weather resistance.
It can be used to formulate chromate-free, water-borne primers to protect metals from corrosion.
Related News: Analogous to master cell lines used to manufacture biopharmaceutical drug products such as monoclonal antibodies, clonal master iPSC lines are a renewable source for manufacturing cell therapy products which are well-defined and uniform in composition, can be mass produced at significant scale in a cost-effective manner, and can be delivered off-the-shelf for patient treatment.2-Acetamido-5-mercapto-1,3,4-thiadiazole Taking Minuo Huawei as an example, in 2016, the capacity utilization rate and production-sales ratio of its multiple drug substance varieties reached more than 90%. Therefore, domestic API companies have expanded their production capacity in recent years to meet the growing demand for APIs.3-Bromo-4-fluorobenzoic Acid At present, Teva can produce more than 300 generic drugs, and the API department has approximately 650 authorized patents and patent applications worldwide. It is also the generic drug company with the most challenges in patenting ParagraphIV in the world.curcumina CAS:458-37-7 It has reserves in the direction of APIs and intermediates for lowering blood lipids, lowering blood sugar and anticoagulation.As part of the agreement, ICIG will enter into a 5-year supply contract to provide Genzyme with materials needed for the production of eliglustat tartrate, an investigational treatment for Gaucher disease Type 1 that is currently in Phase III clinical trials.
| Product Name | |
|---|---|
| 3,4-Dimethyl-1,2-cyclopentanedione | View Details |
| METHYL CYCLOPENTENOLONE Cas:80-71-7 | View Details |
| Propylphosphonic Acid Anhydride | View Details |