


We are 1-Phenyl-1,2,3,4-tetrahydroisoquinoline CAS:22990-19-8 manufacturer and supplier in China, Pls send inquiry of to info@nbinno.com of visit www.nbinno.com our official website should you have any interests
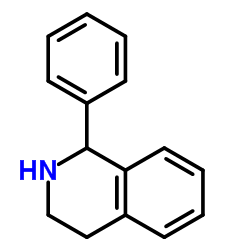
Chemical Name: 1-Phenyl-1,2,3,4-tetrahydroisoquinoline
CAS.NO: 22990-19-8
Synonyms:
1,2,3,4-Tetrahydro-1-phenylisoquinoline
1-Phenyl-1,2,3,4-tetrahydroisoquinoline
1-Phenyl-1,2,3,4-tetrahydro-isoquinoline
Molecular Formula: C15H15N
Molecular Weight: 209.286
Physical and Chemical Properties:
Density:1.1±0.1 g/cm3
Boiling point: 338.4±11.0 °C at 760 mmHg
Melting point:96-98ºC
Flash point: 166.9±14.7 °C
Refractive index:1.589
Specification:
Appearance: White crystalline powder
Purity:≥99%
Packing:
25kg cardboard drum or according to customer specified requirements
Storage:Stored in a cool and dry well-closed container. Keep away from moisture and strong light/heat.
Application:Intermediates of Solifenacin succinate CAS:242478-38-2
Intermediates of Solifenacin CAS:242478-37-1
Related News: With the improvement of people’s living standards and the aging degree, the demand for medicines has been increasing for a long time. Therefore, the transfer from finished medicines to bulk medicines has greatly promoted the demand for bulk medicines.2-Chloro-4-bromo-5-fluorobenzaldehyde “No two studies are the same, and each carries its own unique risks and presents individual challenges, but the aim of CTSuccess is to use the experience we have as a company in supplying materials for thousands of trials to identify the characteristics of a project upfront and highlight potential risk factors that may arise,” said Kristen Devito, Global Director, Catalent Clinical Supply Services.29232-39-1 The foundational patent, which expires in 2034, is owned by MSK and is licensed exclusively to Fate Therapeutics for all human therapeutic uses.21543-49-7 Process: The importance of safety is obvious. The research and development of specialty drug substances (especially high-barrier generic drug substance drugs) usually need to avoid the original process patents, and some chemicals are developed because of the complex structure or the harsh synthetic conditions The synthetic route is more difficult, so the importance of process design capabilities of API companies is becoming more important.Professionally produce and submit DMF files, and supplement data at any time during the patent challenge of formulation companies.
| Product Name | |
|---|---|
| Anisic Acid Cas:NO:100-09-4 | View Details |
| Gadolinium(III) perchlorate Cas:14017-52-8 | View Details |
| 9-bromo-10-(4-phenylnaphthyl-1-yl)anthracene | View Details |