


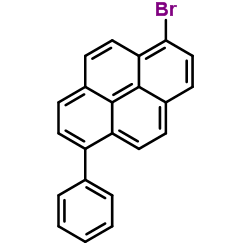
We are 1-Bromo-6-phenylpyrene CAS:294881-47-3 manufacturer and supplier in China, Pls send inquiry of to info@nbinno.com of visit www.nbinno.com our official website should you have any interests
Related News: The program caused the price of some drugs to plunge more than 90% when it was introduced last year in some cities, state news agency Xinhua said.2-(TRIFLUOROMETHYL)PHENYL ISOTHIOCYANATE CAS:1743-86-8 These include: high-barrier generic drug substances and commonly used generic drug substances.4-(2,3-Dichlorophenyl)-5-(methoxycarbonyl)-2,6-dimethyl-1,4-dihydropyridine-3-carboxylic acid Raw material drug manufacturing is listed as one of the top ten key remediation industries, and it is necessary to implement clean transformation, new construction, reconstruction, and expansion of the ten key remediation industry construction projects to implement equal or reduced emissions of major pollutants; ) The industry implements the green enzyme production technology transformation; at the same time, the ten articles of water classify the industrial discharged sewage, and the sewage discharge of API companies is restricted.34966-54-6 These mathematical dosing algorithms were developed in the Damiano Lab at Boston University and refined based on results from home-use clinical trials in adults and children with T1D. Beta Bionics is a Certified B Corporation? whose founders — in addition to Ed Damiano — include other parents of children with type 1 diabetes and people with type 1 diabetes. Beta Bionics is committed to acting in the best interests of the diabetes community and to profoundly disrupting the diabetes medical device industry by bringing the iLet to market as expeditiously and responsibly as possible.INSPIRE is a global, multi-center, randomized, controlled study to assess the efficacy and safety of IV rigosertib in higher-risk MDS (HR-MDS) patients who had progressed on, failed to respond to, or relapsed after previous treatment with a hypomethylating agent (HMA) within nine cycles over the course of one year after initiation of HMA treatment.
| Product Name | |
|---|---|
| Aluminium metaphosphate Cas:13776-88-0 | View Details |
| 4-[2-(2-Amino-4,7-dihydro-4-oxo-3H-pyrrolo[2,3-d]pyrimidin-5-yl)ethyl]-benzoic Acid; PMC Cas:137281-39-1 | View Details |
| germanium oxide Cas:1310-53-8 | View Details |