


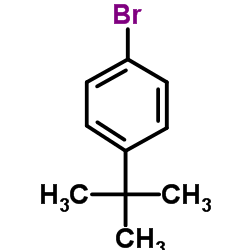
We are 1-Bromo-4-tert-butylbenzene CAS:3972-65-4 manufacturer and supplier in China, Pls send inquiry of to info@nbinno.com of visit www.nbinno.com our official website should you have any interests
Related News: Trial results demonstrated that Oligomannate statistically improved cognitive function in mild-to-moderate AD patients as early as week 4 and the benefit was sustained at each follow-up assessment visit,” Shanghai Green Valley Pharmaceuticals, which developed the drug along with two academic institutions in China, said in a statement.(S)-benzyl 2-(8-amino-1-bromoimidazo[1,5-a]pyrazin-3-yl)pyrrolidine-1-carboxylate Active Pharmaceutical Ingredients (APIs): Pharmaceutical active ingredients, which are the basic substances that constitute the pharmacological effects of pharmaceuticals, and are prepared by chemical synthesis, plant extraction, or biotechnology.ethyl 2-imidazo[1,2-a]pyridin-3-ylacetate CAS:101820-69-3 Active Pharmaceutical Ingredients (APIs): Pharmaceutical active ingredients, which are the basic substances that constitute the pharmacological effects of pharmaceuticals, and are prepared by chemical synthesis, plant extraction, or biotechnology.4-(3-bromophenyl)-6-phenyldibenzo[b,d]furan DMF is the main management method for APIs in developed countries in Europe and the United States. Under the DMF system, API companies can submit DMF filing documents to the regulatory authority at any time, but the regulatory authority will not conduct technical reviews on them. When the drug is administered, the regulatory authority will associate and review the drug substance and the preparation.Only when the drug substance is processed into a pharmaceutical preparation can it become a drug for clinical application.
| Product Name | |
|---|---|
| 12H-Benzofuro[2 3-a]carbazole Cas:1338919-70-2 | View Details |
| 6-Fluoroindole-2-carboxylic acid | View Details |
| Triphenylphosphine | View Details |